|
F S F STEP 2 Total the valence electrons for 1S and 2F. STEP 5 If octets are not complete, form one or more multiple bonds. STEP 4 Add remaining electrons as lone pairs to complete octets (2 for H atoms). STEP 3 Attach the central atom to each bonded atom using one pair of electrons. STEP 2 Add the valence electrons from all the atoms. STEP 1 Determine the arrangement of atoms. The bonding pairs of electrons between atoms. 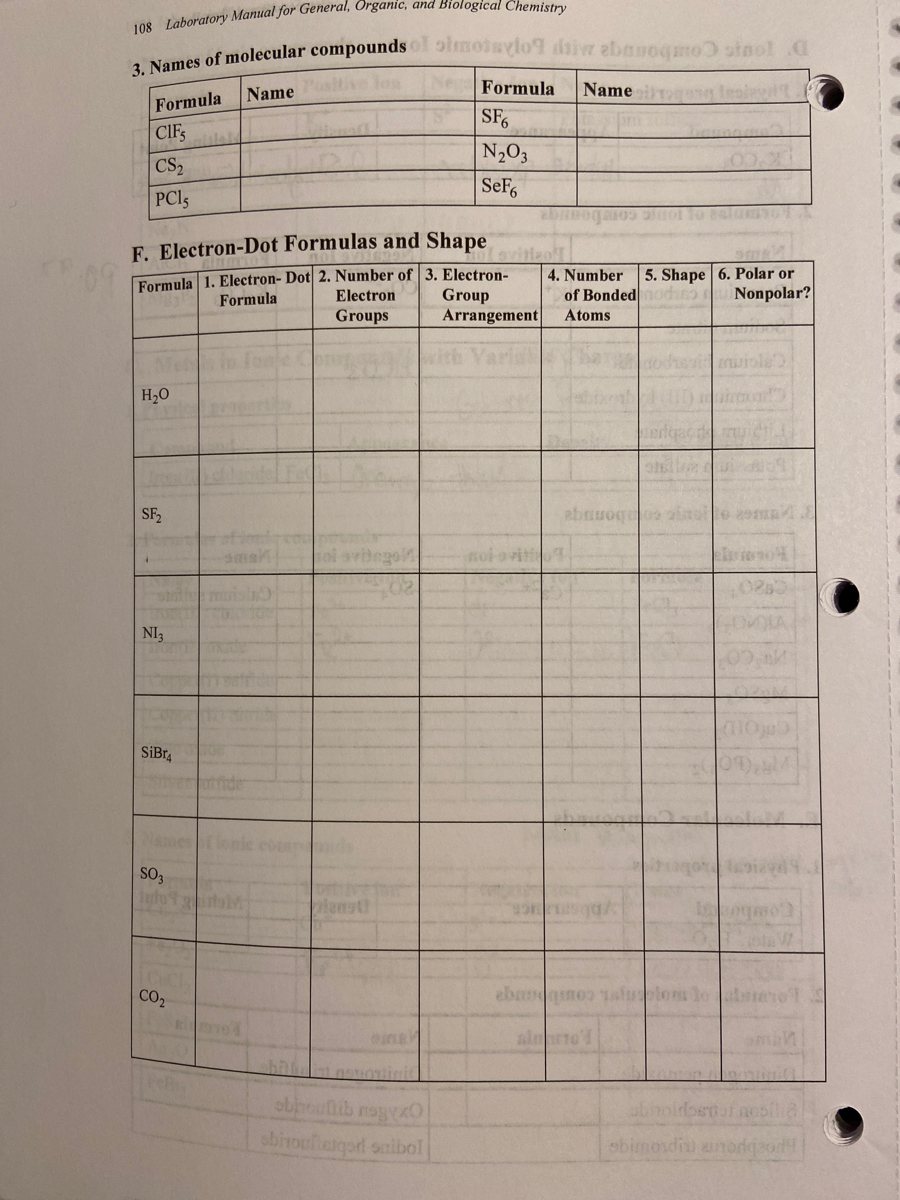
Publishing as Benjamin CummingsĮlectron-dot formulas show The order of bonded atoms in a covalent compound. 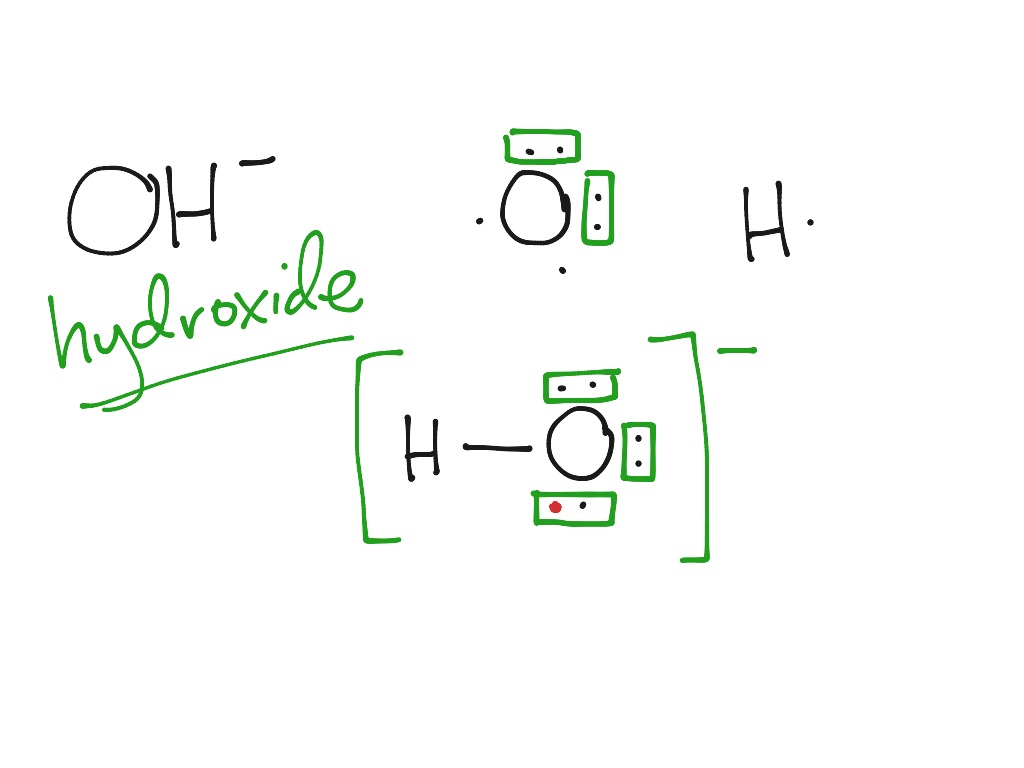 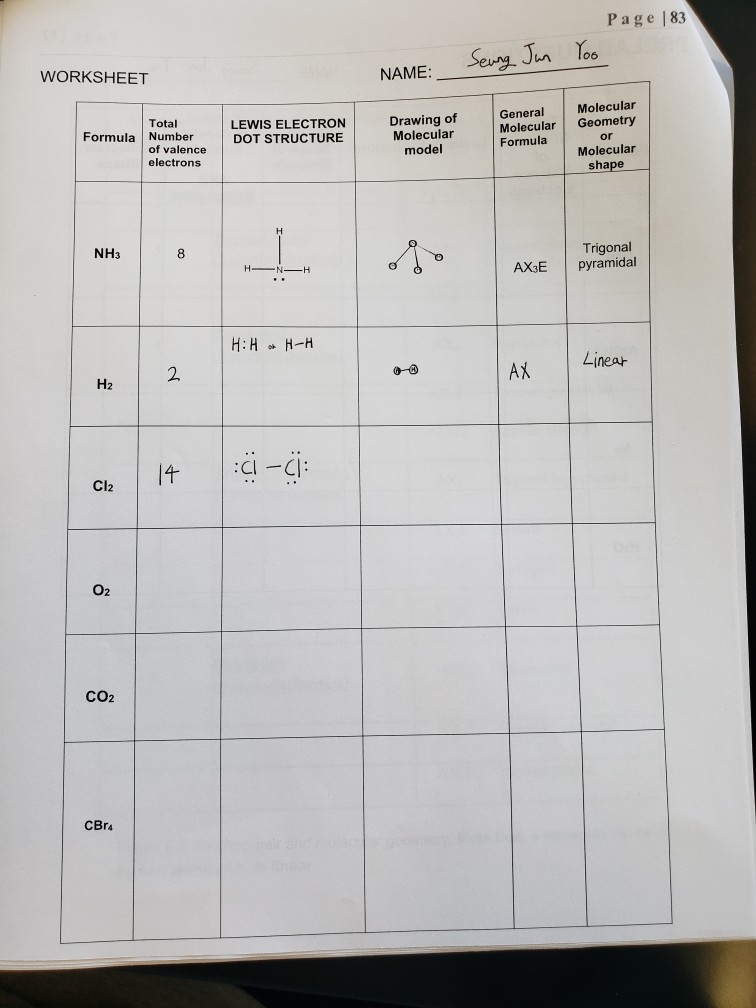
Table 10.1 Copyright © by Pearson Education, Inc. The number of covalent bonds can be determined from the number of electrons needed to complete an octet. 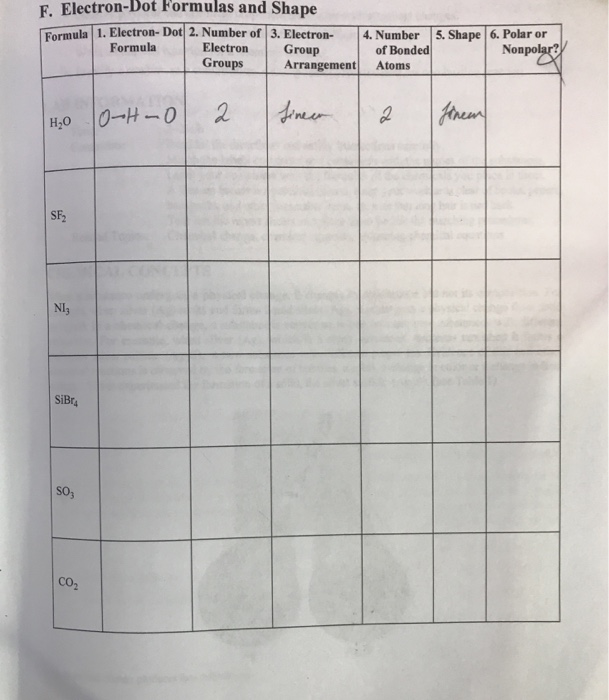
Presentation on theme: "Chapter 10 Molecular Structure: Liquids and Solids"- Presentation transcript:ġ Chapter 10 Molecular Structure: Liquids and Solidsġ0.2 Electron-Dot Formulas Copyright © by Pearson Education, Inc.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
